A Survey of Soil Buffering Capacity in
North Central Massachusetts
by Alec
Andersen, Natalie Brough, Meghan Cameron, Jillian Carkin, Kristi Carroll, Kathleen Craigen,
Shelagh Daly, Valerie DeGroote,
Sasha Donoghue, Molly Flavin,
Ashley Flinkstrom, Lauren Giacobbe,
Thomas Kanan, Christopher Livingstone, Timmy O'Brien
Jr., Devon Picard, Stefani Rivetts, Taylor Sleczkowski, Michael Tenore, and
Daniel Tourtellot
North Middlesex Regional High School, Townsend, MA 01469
Acid rain can have a serious impact on the healthy function of living organisms
(Li, 2006). The more buffer capacity that soil has in lessening the effect of
acid rainfall, the more likely the environment will be to flourish. Our Honors
Earth Science class measured the buffering capacity for several soil samples
that were collected on November 29, 2006 in the
Townsend-Ashby-Pepperell-Fitchburg area.
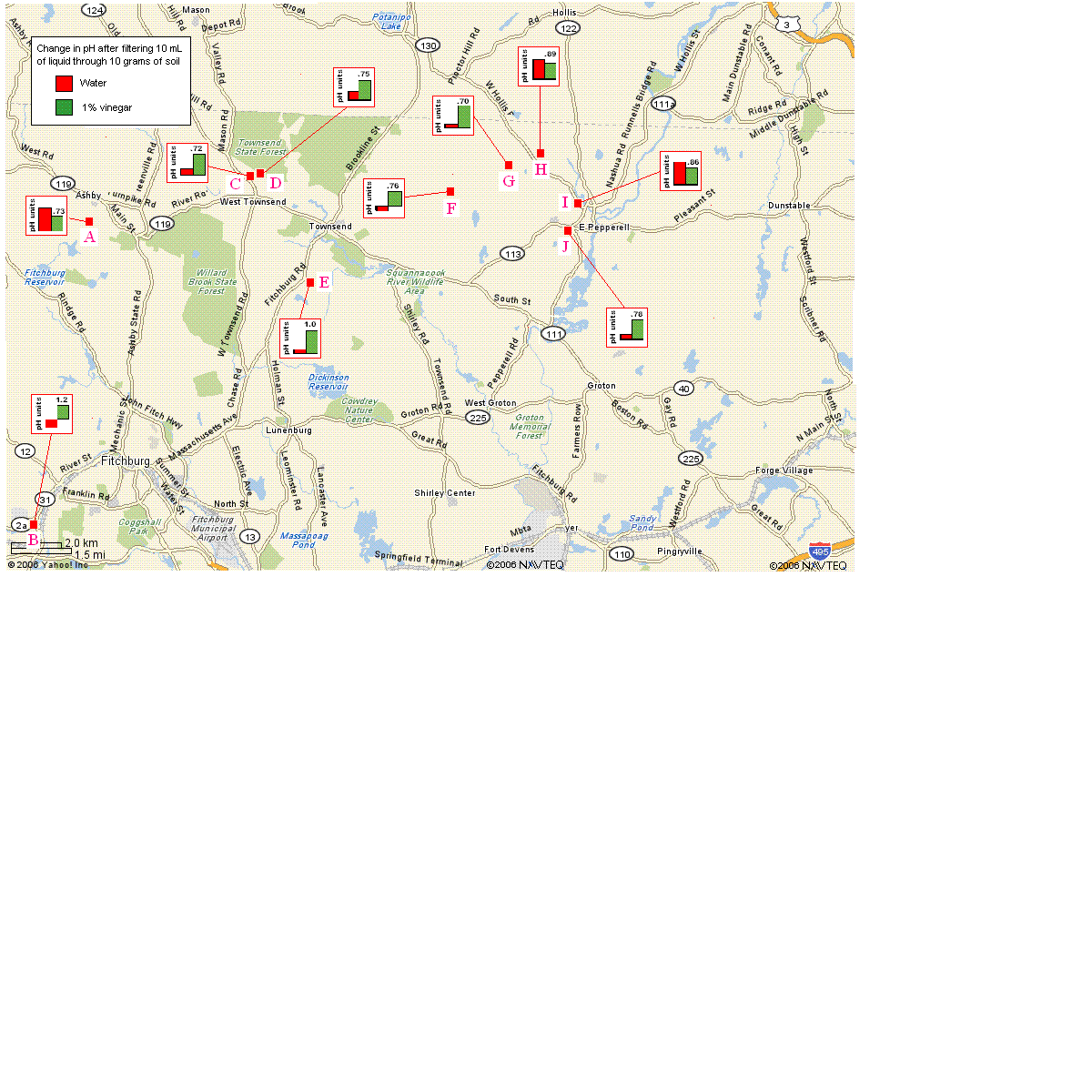
10.0 grams of each soil sample was added to a funnel containing a dampened
piece of filter paper (Ahlstrom, 11 cm diameter). Then we measured the pH of
10.0 mL of tap water before and after letting it
filter through the soil by gravity filtration. pH
readings were measured using Checker pH meters. We then repeated the procedure
using 1% vinegar, a new piece of filter paper, and a fresh 10.0 gram sample of
the same soil.
For all nine of the soils, passing the 1% vinegar through the soil resulted in
an increase in pH of the liquid that was collected ranging from 0.72-1.2 pH units.
Therefore, we can conclude that all soil samples showed some ability to absorb
an acid challenge. For two of the nine soil samples that were tested (Sites B
and F), the pH of the filtered water control was lower than the pH of the water
before being passed through the soil; these sites had more acidic soil than the
other sites. Sites A and I had the least buffering capacity when compared with
the water control.
References
Li, Arthur K. "Environmental Education Site." Education and Manpower Bureau. 24 Nov. 2006. 07 Dec. 2006. http://resources.emb.gov.hk/envir-ed/text/globalissue/e_m2_5_3.htm
Ruesink, Ana, Alan R. Berkowitz, and Kathleen Hogan.
"The Soil Beneath Us." Small
Watershed Ecology Assessment Project. Institute
of Ecosystem Studies (Millbrook NY
12545-0129
http://www.ecostudies.org/images/education/sweap/.%5CMODULE_5.pdf